
Market Opportunity
The market for opioids is estimated to be $30 billion and growing … But the potential for PPL-138 goes beyond that because many people in the world are in severe pain without access to adequate pain control because of fears of addiction liability. Recent research has demonstrated that PPL-138 also has potential for treatment of addiction as well as for treatment of PTSD, thus expanding the market potential further.
Strategic focus
The strategic objective of the company is to partner with appropriate market leader(s) that have the resources to maximize the market potential of the Phoenix compounds. Several leading pharma companies that have a strategic focus on pain or addiction have indicated interest in partnering by the end of Phase 1. We plan to advance PPL-138 into Phase 1 clinical trials In 2025 and then advance it to Proof of Concept in 2026 / 7 in order to position it for optimal deal terms.

Scientific Partners








Growing Market
- Global market estimated at ~$30B Ref 7
- North America > 50% of global demand
- Many people in the world are in severe pain without access to adequate pain control because of fears of addiction liability
- Market can be much larger if addiction risk is removed


PPL-138 Development Timeline
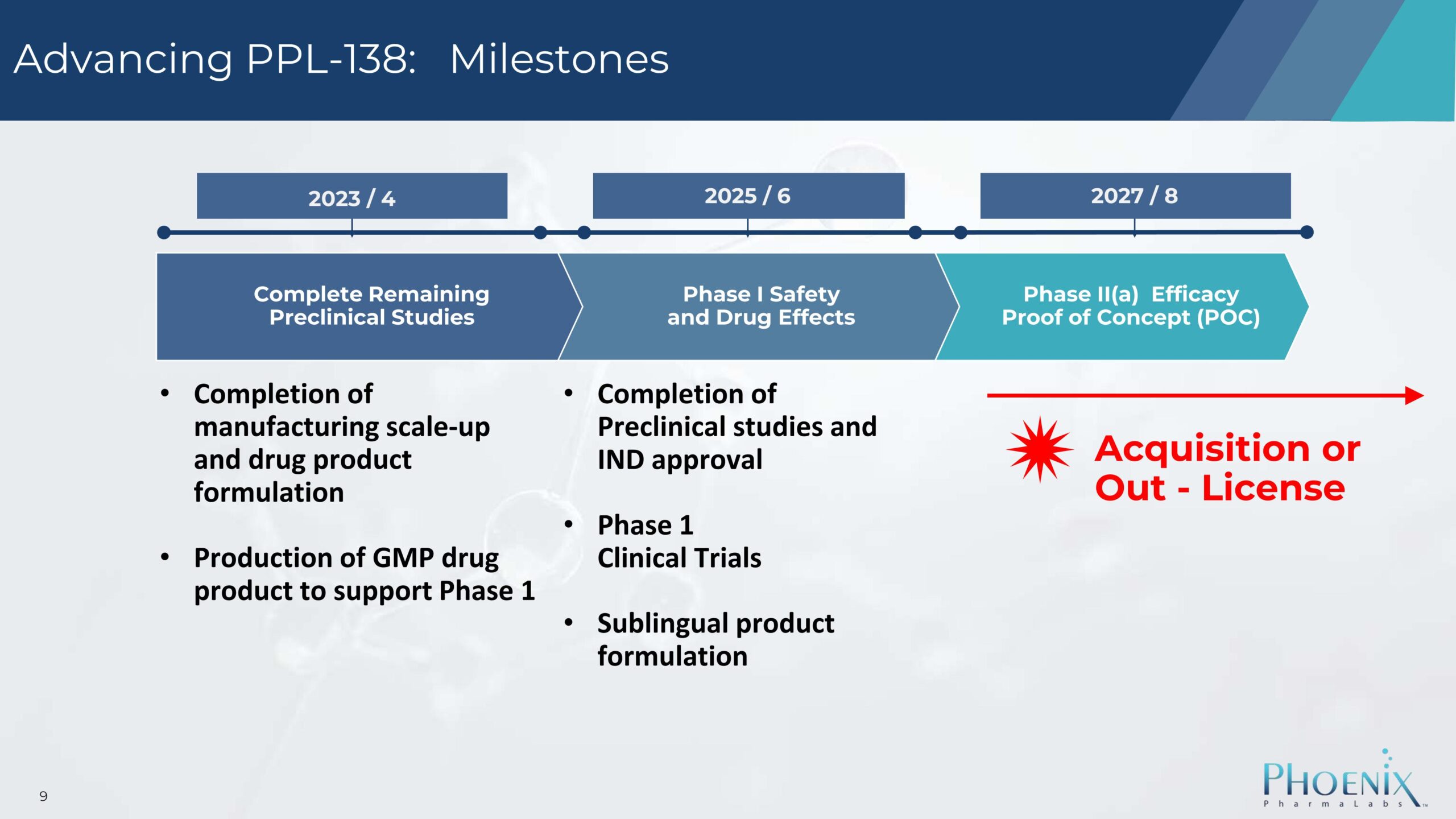
- Completed remaining preclinical studies
- Scaled up manufacturing and finalized drug product formulation
- Produced GMP-grade drug product to support Phase 1 clinical trials
- Complete all preclinical studies and obtain IND (Investigational New Drug) approval
- Begin Phase 1 clinical trials to evaluate safety and drug effects
- Develop a sublingual (under-the-tongue) version of the product
- Begin Phase II(a) clinical trials to test efficacy and establish proof of concept
- Pursue acquisition or out-licensing opportunities based on successful results
About Us
Phoenix PharmaLabs welcomes inquiries by organizations seeking a beneficial relationship. Please contact us to explore potential opportunities.
